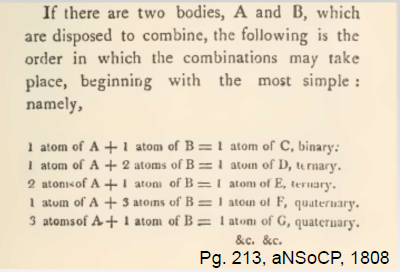Lecture 3
The concept of atoms
DEMOCRITUS (460-370BC) : everything is made of atoms -> indivisible and indestructible
ROBERT BOYLE & ISAAC NEWTON (1600-1700) : Corpuscular theory
LAVOISIER:
ROBERT BOYLE & ISAAC NEWTON (1600-1700) : Corpuscular theory
- No interaction mechanism
- No predictive power
LAVOISIER:
- The elements we know of now are what we can divide and analyse
- No such thing as the ultimate unit (merely fictional)
John Dalton (1766-1844) : atom theory
- made theory of atoms tangible with reality/ experiments
- Hypothesise that 1 atom of H + 1 atom of O -> 1 molecule of water
Dalton's law
- All matter was composed of solid, indivisible and indestructible atoms
- In a chemical reaction, atoms are rearranged.
- Every element has its unique kind of atom. All atoms of an element are identical. Atoms of different element have different weight and properties.
- Dalton's law of multiple proportions: Atoms of different elements combine in whole number ratio
- eg 1 H + 1 O -> HO (Binary system)
Dalton's atomic theory cont.
- Dalton's law of pressure
- Mostly wrong kinetic theory of gases:
- Like atoms repel each other -> partial pressure (but why unlike atoms do not repel each other)
- Different atoms -> different size. Atoms surrounded by caloric. GAS= larger amount of caloric than solid
- -> different atoms have different mass
Dalton's atomic symbol
Some history
- French revolution (1780s-1790s)
- Napoleon (1805)
- Peace in Europe (1815-1914)
- Rapid Industrial revolution(1760-1850s), economic growth
- Colonization
Humphry Davy (1778-1829) - the "celebrity chemist"
- Attempts to describe chemical bonds
- Voltaic pile: 'electropositivity' and ' electronegativity' -> Davy speculates chemical affinity is the result of electrical attraction.
- May decompose some of Lavoisier's 33 elements to simpler substance
- Used electricity to decompose potash -> potassium and soda -> sodium
- Discovered Barium, Strontium, Calcium, Magnesium
Do all acid contain oxygen?
- Lavoisier postulated all acids contain oxygen
- Therefore, HCL(muriatic acid) should also contain oxygen
- Davy tried to burn Chlorine gas (oxymuriatic acid) in carbon to give Carbon oxide-> but failed
- Conclude: this oxymuriatic is not a compound that contain O. Names Chlorine gas.
- Discovered iodine, inferred existence of flourine
- Important for Acid-Base theory
Jons Jacob Berzelius (1779-1848)
Electrical Chemical dualism
- Used VOLTAIC PILE to understand the nature of the constituents of the compounds
- Every chemical compound -> 'electropositive' + 'electronegative' -> combine due to electrochemical affinity
- Chemical reaction -> rearrangement of electropositive and electronegative pair
- Worked very well for chemistry of ionic compounds (INORGANIC CHEMISTRY)
water recognised as H2O in 1805 based on atomic and molecular weights